

Among these collagen is predominant,[3] with established and emerging clinical applications in repairing urogenital damage, skin wounds, bone defects, hernia, cardio-vascular disease, and in surgical reconstruction.[4-9] While the endogenous variety and complexity of decellularized donor tissues (dermis, aortic valves,[10] lung,[11] heart [12, 13] etc.) allow close matching to the needs of a given therapy, this can also be achieved by synthetic bottom-up engineering to allow the complete design of the material.
[3, 14] Advanced manufacturing techniques including 3D printing, electrospinning, moulding, microfluidic encapsulation, acoustic cell-patterning, crosslinking, and composite and modular assembly allow artificial control of degradation rate, structure, and patterning across multiple length scales.[5, 15-19] Through their combination with cell therapy, growth factors, and nanoparticles, unmet needs in spinal cord damage,[20] diabetes,[21] cartilage and connective tissue defects, tooth,[22] and heart regeneration,[17] are now being approached. Beyond collagen, fibrous proteins including silk, elastin and keratin are growing in their use as biomaterials, further expanding the range of material and biological properties available, and the potential for novel therapeutic applications.[5, 19]
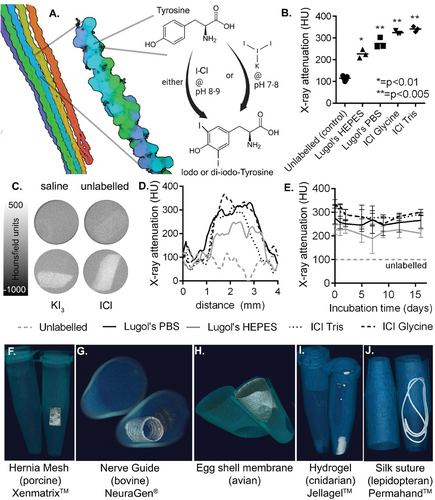
Yet in order for emerging biomaterial therapies to reach maturity, it is crucial to validate their behavior and safety post-implantation.[23-25] However, this task remains challenging due to the inherent resemblance between protein-based implants and native tissue, with both generating similar signals on commonly used medical imaging modalities such as Magnetic Resonance Imaging (MRI), X-ray Computed Tomography (CT), and ultrasound. Consequently, detection of critical events such as successful delivery and integration, mis-delivery, mechanical failure, or detachment proves unachievable in both preclinical models and patient populations, primarily due to the limited availability of imageable scaffolds. Though both X-ray visible collagen and silk materials have been previously developed, these have suffered either from a reduction in material strength due to labeling,[26] or the low practicality of stably and homogenously incorporating nanoparticle-based contrast agents into pre-formed scaffolds such as decellularized tissue.[27]
Though various chemistries are compatible with whole collagen scaffolds including aldehyde or carbodiimide-based reactions for crosslinking,[3, 6] conjugation of cell attachment sites, and introduction of biochemically active groups,[28, 29] there has been little focus on enabling non-invasive in vivo imaging.